




Resonant Transmutation of Sodium in Human Sweat
Sweat Gland Secretions and the Sodium-Potassium Balance
by Alex Putney for Human-Resonance.org
January 25, 2013 -- Updated April 17, 2017
The optical transmission of infrared light through the body's meridian system stimulates the flow of light-storing DNA granules to rapidly and effectively rebalance the luminal field of the human body. Moxa treatments provide direct infrared illumination to selected acupressure points, bringing highly localized skin surfaces to temperatures as high as 55°C for just a few seconds, before brief withdrawl of the heat (Li et al, 2009).
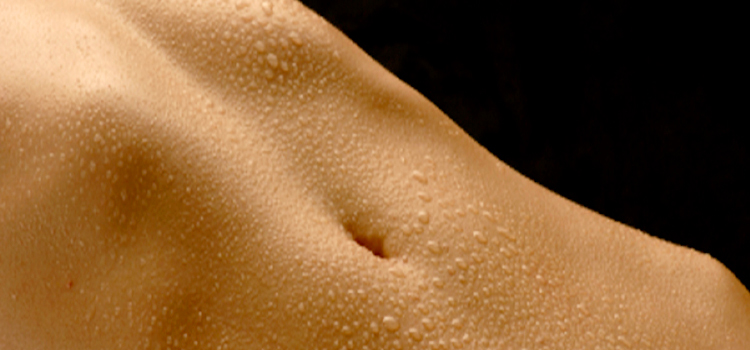
Skin temperatures during rest periods measured at different points of the body show a normal range of 19-29°C (Benedict, et al., 1919). Relatively recent discoveries in the field of thermography, or the visualization of infrared light, have been widely applied in diagnosing animal or human bodies for fungal infection and cancer diagnosis, also allowing direct imaging of distinct temperature variations observed between the warmer areas of the body where lymph node clusters and glands tend to be located, as opposed to the colder nasal region, the extremities and areas where excess fat is stored below the skin.
When properly applied, intense infrared and ultraviolet-A light elevates skin metabolism into a heightened state that alters the elemental makeup of sweat gland secretions. The fascinating details of this heat-induced mode of enhanced skin metabolism were first quantified by C.L. Kervran in cases of heat stress of workers during oil prospecting activities in the arid conditions of North Africa's Sahara desert in 1959.
To determine necessary dietary enhancements for the continually heat-stressed workers, Kervran created a closed environment to study the unusual relationship between sodium and potassium in human sweat. In total, 808 analyses were conducted to carefully quantify the elemental makeup of all liquids and solids ingested and excreted by the workers, even including sponging off sweat secretion samples. Test results confirmed that the ratio of potassium to sodium in the excreta of workers increased with temperature.
A full quarter, or an average of 1.28g per day, of ingested sodium could not be accounted for in their wastes. If this sodium was simply being stored in the body, a six-month accumulation would amount to a kilogram, suggesting that sodium was being converted into another element altogether. Painstaking analyses confirmed that the missing sodium was excreted as excess potassium, that otherwise could not be accounted for. Furthermore, data revealed that seasonal temperature increases over the course of the study from May to September, showed a clear correlation with the changes in the potassium to sodium ratio (K / Na) of workers' perspiration. This was unmistakable evidence for biological transmutation.
Kervran's report was published in 1963, presenting carefully drawn conclusions that comprised the identification of resonant nuclear transmutations occurring in the human body. He suggested a crucial role of oxygen in this temperature-dependent reaction by showing that oxygen possesses the exact atomic mass disparity between sodium into potassium. However, conversion reaction is far more complex.
Subsequent attempts to replicate this reaction in vitro were successful, and progessed to more complex experimental setups in Japanese universities, where the reaction was replicated under spectrographic observation by various other researchers using sodium vapor (Torii, Sakurazawa, Odagiri, 1963; Ohsawa, Kushi, 1964). The spectral emission lines of potassium consistently appeared in the pure sodium vapor.
Time and time again, this reliable experiment revealed the same startling result --sodium vapor heated at low temperatures converted into potassium at a stable rate that increased with electrical input to the system. Neon and argon gases added to the sodium/oxygen reaction were found to increase the potassium output, for unknown reasons. Phonon resonance calculations determine precise temperature thresholds that induce atomic conversion cascade chains involving neon and argon gases:
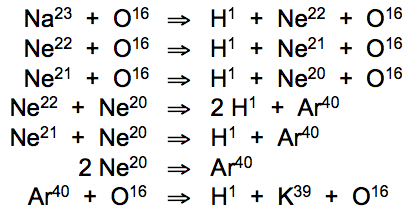
Phonon frequency matching of oxygen heated to 37.8°C with hydrogen at rest (20°C) induces conversion of sodium atoms into neon isotopes (Ne22, Ne21, Ne20). Phonon resonance of Ne20 atoms at 37.7°C with stable argon isotope (Ar40) induces resonant fusion of two neon gas atoms into a single argon gas atom. O/H resonance at 37.8°C then induces fission of argon atoms, yielding hydrogen and potassium (K39).

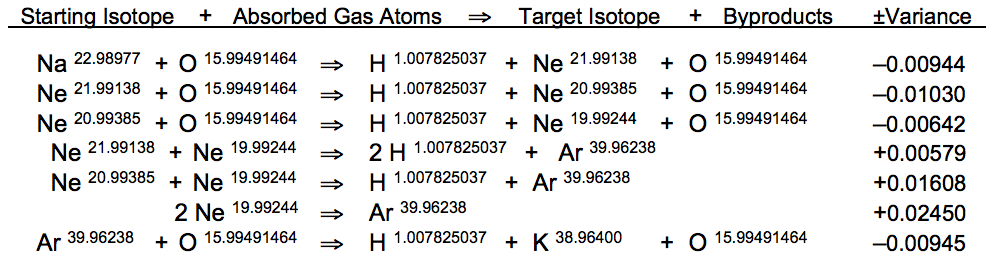
The ± variance in atomic weight between starting isotope and absorbed gas atom in relation to the target isotope and reaction byproducts differs significantly among these atomic conversions. As typifies resonant nuclear reactions taking place in the bloodstream, oxygen-based reactions in the Na => K sequence display a negative variance (starting isotopes weigh less than the products) while the 2 Ne => Ar reactions display a positive variance (starting isotopes outweigh the products). During the 2 Ne20 => Ar40 fusion reaction, an excess atomic mass of 0.02450 U is ejected as an electron pair, comprising 0.0010971598 U, while the remaining 0.0234028402 U is emitted as photons; primary contributors to the ultra-weak biophoton radiation of human skin. Fluctuations in emission intensity from skin are caused by variations of planetary infrasound.

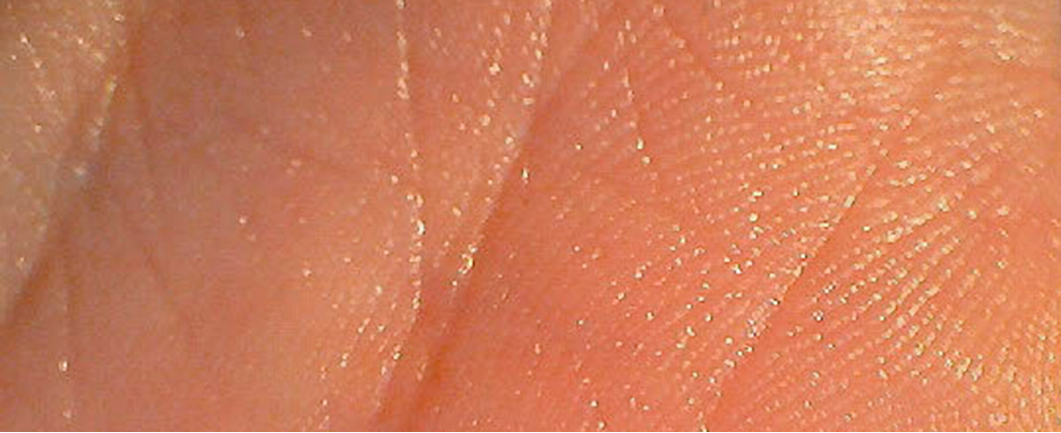
Scanning electron microscopy of the dermal ridges of human hands and fingerprints have captured detailed images of individual pores of the sweat glands and their tiny sweat secretions (above). Shown in a heightened relief, the configuration of pores provides a unique fingerprint that leaves its watermark in the world as traces of sweat are perpetually secreted (below). Forensic science has been rapidly advanced using fingerprint, skin, hair and saliva samples for DNA pattern analyses to identify criminal suspects, and the present applications of phonon resonance physics similarly identifies unique patterns in the fundamental isotopic signatures of the elements that enable all biological metabolic processes.

Copyright 2013-2017 Alexander Putney