




Resonance in the Geyser Reactor System
The Geyser Reactor Transmutation System
by Alex Putney for Human-Resonance.org
August 13, 2014
Great inventions often display a natural simplicity inspired by keen observations of the surrounding environment --meteorological, geological or biological. The remarkably simple Geyser Reactor system completed by Alexander Putney in 2014 (below) decisively refutes many long held misconceptions of geology, biology and atomic physics by clearly defining and demonstrating the set of low energy conditions that induce resonant nuclear transmutation.

The Geyser Reactor process represents an unexpected bi-product of twelve years of focused study of ancient psychoacoustic instruments and architectural elements tuned for biorhythmic synchronization. Controlled frequency resonance was also applied by ancient cultures in advanced metallurgical techniques enabling induction of atomic transmutation reactions by phonon resonance relationships of metals and absorbed gas atoms.
Replicating hydrological transmutation, the Geyser Reactor maintains a cyclical two-phase process for conversion of various metals as ultrafine powders. This digitally-controlled system drives a basic two-stage reaction that applies newly defined physical laws of resonant atomic transmutation that account for the abundance of gold and other precious metals within hydrological steam vents and geysers. Volcanic hotsprings are now known to accumulate precious metals by perpetuating nuclear transmutation reactions of base metal particles with volcanic gases absorbed at high temperatures and pressures during prolonged periods within fractured bedrock chambers, suffering periodic eruptions that effectively quench the hot metals as they emerge from geysers at the surface.

Carbon dioxide-producing active yeast reactors developed by Joe Champion in 2001 applied phonon resonance formulae, as first conceived by geologist Walter Lussage in 1967, for biological transmutation of silver surfaces to nanogold (above). Now concisely explained within the context of geological and biological transmutation regimes, essential atomic conversion processes are now achievable in simpler digitally-controlled abiotic phonon reactors requiring only heated water and carbon dioxide nanobubbles to convert bulk silver nanopowder into pure gold.
Kaneo Chiba of Reo Lab. Co. demonstrated production and application of nanobubbles in various processes, including bulk waste treatment, food sterilization and preservation with ozone nanobubbles, in addition to health enhancement of most aquatic and terrestrial organisms exposed to oxygen nanobubbles. Initial processes for nanobubble production involved cavitation with ultrasound, yet simple carbon-based ceramic nozzles developed by Satoshi Anzai of Anzai Kantetsu Co. presented an extremely cost effective production method in 2014:

As the main component of this class of carbon-ceramics, amorphous carbon particles (SEM above) contribute micron-sized pores to the composite material that allow passage of gas under low pressure through the nozzles to generate micron-sized bubbles from submerged nozzle surfaces. In still water, microbubbles rapidly coalesce to form larger bubbles that cannot remain suspended in the liquid medium but escape to the water's surface.
However, investigation of the behavior of microbubbles produced from the carbon-based ceramic surfaces into a narrow jet of fast-flowing water revealed several surprising phenomena (below). The rapid motion of microbubbles torn away from nozzle surfaces begins a process known as adiabatic compression leading to collapse by isothermal evolution, whereby reducing in size during the course of several minutes to form nanobubbles (Ohgaki et al., 2010).

During the microbubbles' decrease in size due to surface tension effects driving the dissolution of interior gases into the surrounding liquid, reactive oxygen species (ROS) are generated that decompose organic chemicals and contribute to the beneficial breakdown of toxins within biological systems and the natural environment. Stabilization as long-lived nanobubbles occurs when ions bind to the gas/liquid interface, yet display full collapse and complete dissolution after several minutes in the absence of bound ions.
Longterm studies of the longevity of gas nanobubbles stabilized at <200nm in size in bottled water samples have shown their presence in significant quantity several months after the infusion and bottling process (Takahashi, 2005). Nanobubble stabilization is also influenced by repulsive electrostatic forces due to surface charging, and may be maintained over long periods in colloidal suspensions of silver nanoparticles. Studies of nanoscale forces and fluid/gas dynamics reveal many surprising properties that contribute significantly to our understanding of basic metabolic processes that determine the cellular health of living organisms and entire ecosystems.
Use of carbon-based ceramic nozzles with Geyser Reactor transmutation systems confirms nanobubble technology to represent a cost-effective solution for the efficient bulk delivery of carbon dioxide gas for binding with silver nanoparticles and rapid absorption under ambient pressures. Bulk binding of gas nanobubbles with metal nanoparticles replicates the metabolic activity of hemoglobin in red blood cells, enabling high gas absorption and bulk transmutation rates that far exceed those associated with resonant transmutation in healthy organisms, even under bioelectrification conditions that increase absorption of gases by metals within the body's tissues.
A compact device for producing gas nanobubbles remains the only component of the Geyser Reactor system not readily available, requiring fabrication from special gas permeable materials. While the carbon-ceramic nozzles developed and demonstrated by Anzai Kantetsu represent cost-effective alternatives to high-pressure, high-temperature cavitation machines for nanobubble production, their new nozzles are not yet available for order.
Carbon-ceramics are manufactured by wet packing 60% carbon, 40% clay powder mixtures into nozzle molds before drying and firing @ >1000°C in a reducing or inert gas atmosphere. Viable carbon-ceramics that allow the passage of gas through micropores are commonly used for high-temperature glass and metal casting applications, and can be easily fabricated into a nozzle by reshaping carbon-ceramic mold materials into the desired form.

However, an even simpler nanobubble device has been integrated into the design of the Geyser Reactor system, consisting of a pyrolyzed segment of hardwood tree branch that maintains the natural nanoarchitecture of living wood, generally referred to as 'biological charcoal'. The natural nanopiping of tree wood employs surface wetting effects for pumping water up to the leaves, sometimes hundreds of feet into the sky, yet also facilitates production of nanobubbles. Electron microscopy reveals the complex nanostructures of biocharcoal, presenting networks of carbon nanotubes arranged lengthwise in concentric rings with interconnecting nanopores (SEM above).
Biocharcoal nozzles offer the same basic nanobubble characteristics demonstrated by the carbon-ceramics of Anzai Kantetsu --at a much lower cost of just 25¢ per nozzle-- reflecting the simple natural solutions of Ayurveda. By carefully selecting and tooling the surfaces of a pyrolyzed hardwood branch segment that has no large pores or cracks, and sealing the central channel which tends to be much larger than the nanotubes arranged in concentric rings, an extremely cheap alternative can be produced with minimal cost and effort, in any part of the world.

The ideal nozzle form is bullet-like to allow efficient waterflow over its surfaces (above). Biocharcoal can be quickly reduced with abrasive sandpaper down to the diameter of tubing to be used for delivering low pressure gas flow. Cracking is the biggest obstacle in this basic production process, causing structural losses within the wood, and requires slow, thorough drying of selected hardwood branch segments, followed by heating for 25 minutes @ ~250°C in a reducing or inert gas atmosphere. Size and shape of the nozzle can be adjusted to fit any usable segment of biocharcoal, provided the finished nozzle can be fitted into the pressure tubing securely.
Putney's basic Geyser Reactor transmutation systems operate within the well-known thermoregulation ranges maintained by a healthy human body to achieve resonant conversion of zinc to copper by the same processes that rebalance blood metals within the red blood cells of all mammal species. This simple circulatory heating and cooling process can also be applied under identical conditions to achieve resonant nuclear conversions of gold to platinum and silver to palladium. Exact resonant atomic mass recombination patterns are provided below:
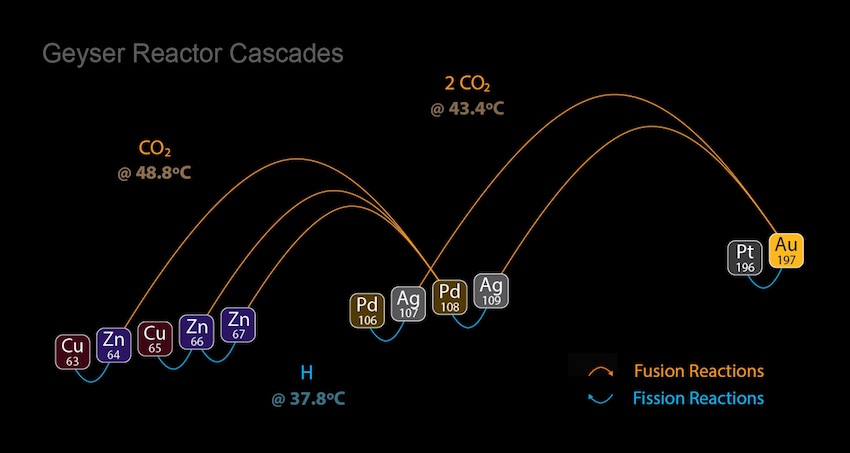
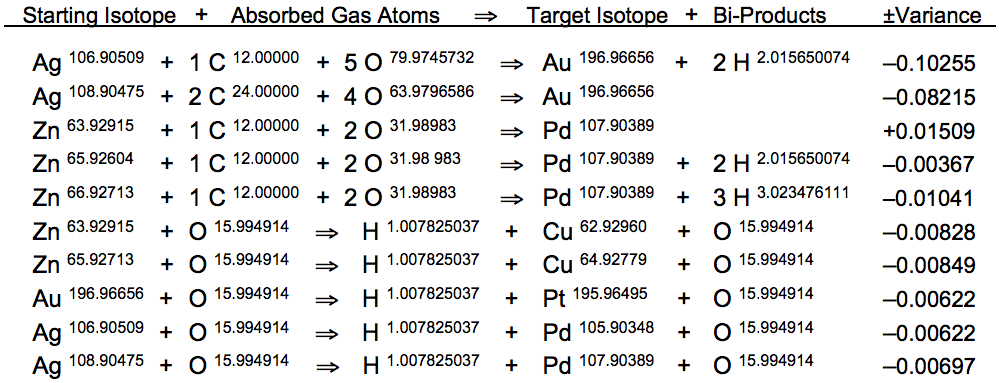
The set of hydrogen-producing nuclear fission reactions occurring at 37.8°C is complemented by another pair of transmutation reactions following a different atomic mass recombination pattern dependent on carbon dioxide gas, enabling more profitable fusion reactions for converting silver to gold at 43.4°C, as well as zinc to palladium at 48.8°C. The atomic mass of CO2 gas precisely corresponds to atomic mass disparity between Ag109 and Au197, as well as Zn64 and Pd108. These confirmed nuclear reactions display a mean atomic mass variance of --0.02365u.
A new resonant class of safe nuclear transmutation systems exemplified by the Ecat reactor of Dr. A. Rossi must inevitably replace the many radioactive industrial atomic power plants that presently endanger all life on Earth.
The extreme scientific ignorance obfuscating the reality of 'cold fusion' demands the free distribution of this major discovery as an open source technology. Geyser Reactors of all sizes can be safely built and operated by students of geology and biology, inspiring advanced metallurgy projects for production of precious metals and superalloys.
Geyser Reactor systems represent the simplest means for safe and efficient nuclear conversion of various metals near room temperature and under ambient atmospheric pressure conditions. The mnemonic phonon physics of resonant atomic transmutation, comprehensively defined here for the first time, now enables more complex high-temperature furnace processes involving conversion of metals by rapid quenching in water after prolonged dwell times at resonance in a molten state. Experimentation with copper bars in standard furnaces confirms that enhanced gas absorption at higher temperatures facilitates conversion of copper surfaces into nickel during cooling in air to 37.8°C by the same hydrogen fission reaction that defines the specific thermoregulation range of the human body.

In geological contexts, silver and gold are typically associated with quartz veins formed during hydrological venting processes occurring below geyser hotsprings such as those spectacular examples located in Yellowstone National Park. Close interaction of lava chambers with overlying aquifers and geysers has led many geologists to the basic hypothesis that precious metals are transported from below by upwellings of superhot, highly-mineralized waters.
If resonant transmutation occurs at that stage, wouldn't there be evidence of the commingling of elements within both silver and gold deposits? In fact, such evidence does exist, but is very rare. Raw gold nuggets do not contain remnants of silver atoms because transport into fractured bedrock piping recurs during multiple explosive events that liquefy the metals and allow their separation by weight differential. Being almost double the atomic mass of silver, liquefied gold atoms readily reconsolidate and part from silver under such extreme conditions.

Beautifully colored thermophilic algae thrive at 46°C in hydrothermal vents like Fly Geyser, located in Gerlach, Nevada (above), reflecting the same microbial abundance that mesmerizes vulcanologists studying seafloor vent processes that support diverse biological communities congregating around towering deepsea 'black smokers'.
Direct evidence for the natural formation of atomically commingled Ag/Au alloys is present within both biological and geological contexts, as yeast species employ the same gas at the same temperatures that affect conversion in geyser vents. This previously unrecognized class of evidence has been described as a yellowish form of silver dubbed 'allotropic silver' that is actually an atomically commingled product of resonant nuclear transmutation of silver into gold. The yellowish complexion of 'allotropic silver' is actually a dispersed coating of gold nanodots comprising <5% of silver surfaces, now rapidly reproduced in digitally controlled abiotic Geyser Reactor systems.
From the book The Geyser Reactor
Continue with Silver into Gold Transmutation
Return to Quantum Trappings
Copyright 2013-2015 Alexander Putney